Why is neon in period 2?
 Sebastian Wright
Sebastian Wright 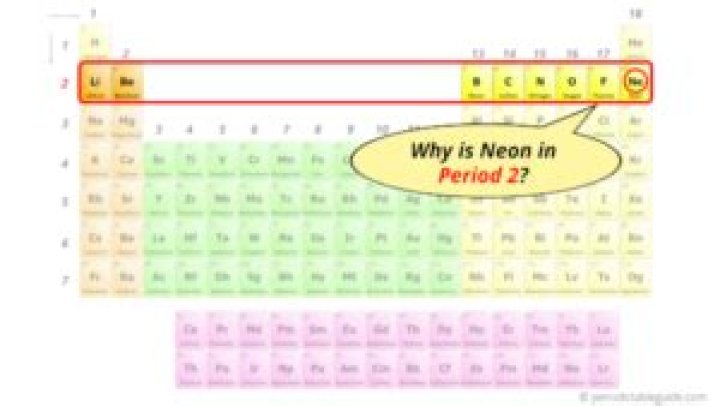
Period 2 elements (carbon, nitrogen, oxygen, fluorine and neon) obey the octet rule in that they need eight electrons to complete their valence shell (lithium and beryllium obey duet rule, boron is electron deficient.), where at most eight electrons can be accommodated: two in the 2s orbital and six in the 2p subshell.
Is neon an 18 period?
Neon (Ne), chemical element, inert gas of Group 18 (noble gases) of the periodic table, used in electric signs and fluorescent lamps.
What group or period ends with neon?
noble gases
The six noble gases—helium, neon, argon, krypton, xenon, and radon—occur at the ends of the six completed periods and constitute the Group 18 (0) group of the periodic system.
When was neon founded?
1898
Neon was discovered in London in 1898 by a pair of British chemists: Sir William Ramsay and Morris W. Travers. Ramsay chilled a sample of air until it became a liquid. He then warmed this liquid, capturing the gases as they boiled off.
What are 5 uses for neon?
Neon is used in vacuum tubes, high-voltage indicators, lightning arresters, wavemeter tubes, television tubes, and helium–neon lasers. Liquefied neon is commercially used as a cryogenic refrigerant in applications not requiring the lower temperature range attainable with more extreme liquid-helium refrigeration.
What are 3 uses of neon?
Neon: uses
- used in making neon advertising signs, which accounts for its largest use.
- used to make high-voltage indicators, lightning arrestors, wave meter tubes, and TV tubes.
- neon and helium are used in making gas lasers.
- liquid neon is an economical cryogenic refrigerant.
What are 5 interesting facts about neon?
Quick Facts about Neon
- Neon is a noble gas. It’s colorless and has no smell.
- Neon glows a reddish-orange color when put in a tube.
- Neon is one of the rarest elements on Earth.
- Scientists Morris W. Travers and Sir William Ramsay discovered Neon in 1898.
- Neon is commonly used in lights that are known as neon signs.
What is the period number of oxygen?
Fact box
Group 16 −218.79°C, −361.82°F, 54.36 K Period 2 −182.962°C, −297.332°F, 90.188 K Block p 0.001308 Atomic number 8 15.999 State at 20°C Gas 16O Is neon shiny or dull?
Element Luster Malleability Neon Colorless Not able to be stretched Silicon Dull, metallic Brittle, shatters when shaped Sulfur Dull Cannot be stretched Manganese Shiny Can be hammered into thin sheets What do you need to know about the element neon?
Neon is a noble gas element. Trivial name of Neon is noble gases*, aerogens. Know everything about Neon Facts, Physical Properties, Chemical Properties, Electronic configuration, Atomic and Crystal Structure. Neon is a chemical element with symbol Ne and atomic number 10.
How many electrons does neon have on the periodic table?
Neon is a 10. chemical element in the periodic table of elements. It has 10 protons and 10 electrons in the atomic structure. The chemical symbol for Neon is Ne.
What is the atomic weight of NE neon?
Ne Neon – Element Information, Facts, Properties, Trends,Uses, Comparison with other elements. Element 10 of Periodic table is Neon with atomic number 10, atomic weight 20.1797. Neon, symbol Ne, has a Face Centered Cubic structure and Colorless color. Neon is a noble gas element. Trivial name of Neon is noble gases*, aerogens.
Who was the first person to discover neon?
Neon was discovered by Sir William Ramsay, Morris W. Travers at 1898 in London, England. There are three naturally occurring isotopes of neon exist, 20 Ne, 21 Ne and 22 Ne.