What is the n value of AlCl3?
 Sebastian Wright
Sebastian Wright 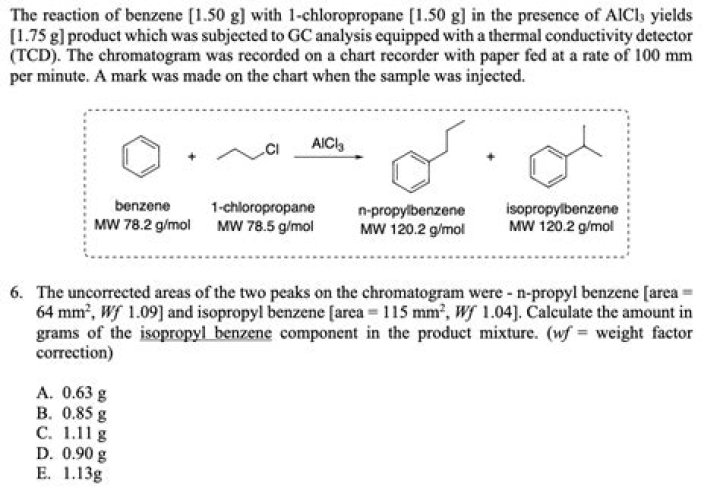
Aluminium chloride (AlCl3), also known as aluminium trichloride, describe compounds with the formula AlCl3(H2O)n (n = 0 or 6). They consist of aluminium and chlorine atoms in a 1:3 ratio, and one form also contains six waters of hydration.
What is the charge of Al in AlCl3?
+3
Al has a charge of +3. Cl has a charge of -1. Hence 3 chloride ions are required to neutralize charge of +3 on aluminium and the compound formed is AlCl3 . Three chloride ions are required to bond with one aluminum ion.
What is AlCl3 used for?
Industrial Uses of Aluminium Chloride (AlCl3) Aluminium chloride is used widely in manufacturing rubber, lubricants, wood preservatives, and paints. It is used in pesticides and pharmaceuticals. As a flux in Aluminium melting. It is used as antiperspirant.
What is the name of two AlCl3?
Aluminum chloride is a chemical compound with the chemical formula AlCl3. When contaminated with iron chloride, it often displays a yellow color compared to the white pure compound. It is used in various chemical applications as a Lewis base, with anhydrous aluminium trichloride being the most commonly used Lewis acid.
Is AlCl3 acidic or basic?
No, it means AlCl3 is a weak acid, because the only part of the molecule that is contributing to the pH of the solution is the Al+3 ion. The conjugate base of a strong acid has very little impact on the pH of the solution (with the case of Cl- and its conjugate acid, HCl).
Is AlCl3 a Lewis acid?
Aluminum chloride (AlCl3) is a Lewis acid because the aluminum atom has an open valence shell. When aluminum chloride is under discussion it is called a Lewis acid or an electrophile.
Does AlCl3 have a formal charge?
neutral formal charge). AlCl3 dimerises because of the unfilled octet in the aluminium.
Which acid is used in AlCl3?
As Al of AlCl3 can accept a lone pair it is called Lewis acid.