What happens when you mix oxygen and hydrogen?
 Robert Bradley
Robert Bradley 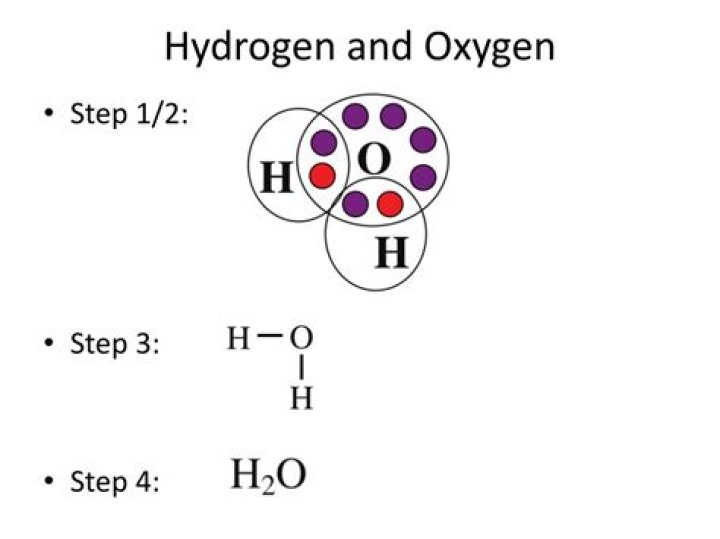
When molecular hydrogen (H2) and oxygen (O2) are combined and allowed to react together, energy is released and the molecules of hydrogen and oxygen can combine to form either water or hydrogen peroxide. For both of the reactions shown, the hydrogen molecules are oxidized and the oxygen atoms are reduced.
What type of reaction is hydrogen and oxygen?
Exothermic Reaction Like all fuels, the reactants, in this case hydrogen and oxygen, are at a higher energy level than the products of the reaction. This results in the net release of energy from the reaction, and this is known as an exothermic reaction.
What is hydrogen and oxygen as a compound?
Water, which is a chemical compound of hydrogen and oxygen in the ratio two hydrogen atoms for every oxygen atom, contains H2O molecules.
Why does it take 2 hydrogens to combine with 1 oxygen to make water?
Oxygen is usually present as O2 (two oxygen atoms bonded together), so in order to make water (H2O) the two oxygen atoms in O2 must break apart, and that takes extra energy. This energy can come from a couple places.
Why does oxygen and hydrogen bond?
Covalent bonds occur when two atoms—in this case oxygen and hydrogen—share electrons with each other. Because oxygen and hydrogen attract the shared electrons unequally, each end of the V-shaped H2O molecule adopts a slightly different charge.
What is the difference between a bottle of water and a bottle of hydrogen and oxygen gas?
The difference is due to the fact that one water molecule has two hydrogen atoms to one oxygen atom, as explained above. This means it takes two water molecules (2 H2O) to make one oxygen molecule (O2). At the same time, however, two molecules of water (2 H2O) can make two molecules of hydrogen (2 H2).
What is the chemical formula of oxygen?
O₂
Dioxygen/Formula
What is the chemical formula of H O?
Hydroxide
| PubChem CID | 961 |
|---|---|
| Structure | Find Similar Structures |
| Molecular Formula | HO- |
| Synonyms | hydroxide hydroxide ion hydroxyl ion oxidanide Hydroxyl anion More… |
| Molecular Weight | 17.007 |
Can na2o absorb oxygen?
The octahydrate is produced by treating sodium hydroxide with hydrogen peroxide. Sodium peroxide can be prepared on a large scale by the reaction of metallic sodium with oxygen at 130–200 °C, a process that generates sodium oxide, which in a separate stage absorbs oxygen: 4 Na + O2 → 2 Na2O.
Can water be created artificially?
Is it possible to make water? Theoretically, it is possible. You would need to combine two moles of hydrogen gas and one mole of oxygen gas to turn them into water. However, you need activation energy to join them together and start the reaction.
Why do we have two hydrogens in oxygen?
Oxygen needs two electrons to fill its outermost electron shell. Hydrogen has one electron to play with. Therefore, an oxygen atom needs two hydrogens to bond to in order to complete its shell.
Which is element makes dihydrogen oxide ( H2O )?
Hydrogen and oxygen. What is probably meant is which two elements make dihydrogen oxide (water; H2O). The answer remains the same: hydrogen and oxygen. What is hydrogen plus oxygen? Hydrogen plus Oxygen can make many different substances. For example, H2O is water. But H2O2 is hydrogen peroxide. Can you make water from hydrogen?
How does hydrogen and oxygen bond to form water?
By forming H 2, the atoms can share electrons to achieve full 1s orbitals. Oxygen has more electrons than hydrogen, but oxygen atoms are two electrons short of having a filled 2p orbital. Bonding to form O 2 allows them to share electrons and fill the 2p orbitals. To form water, an oxygen atom bonds with two hydrogens.
What happens when oxygen and water are combined?
Oxygen is made of a oxygen atom and water is made out of a oxygen atom and two hydrogen atoms. When combined under the right conditions, hydrogen peroxide can be formed (H2O2).



